
Protein Chromatography: Process Development and Scale-Up, 2nd Edition
Price: $ 65.99
4.8(706)
An all-in-one practical guide on how to efficiently use chromatographic separation methods Based on a training course that teaches the theoretical as

Frontiers 1Progress, applications, challenges and prospects of protein purification technology
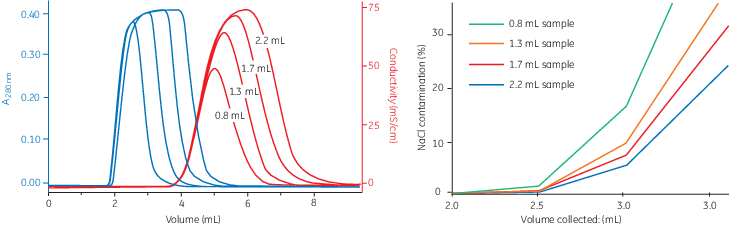
Size Exclusion Chromatography

Chapter 3: Investigating Proteins - Chemistry
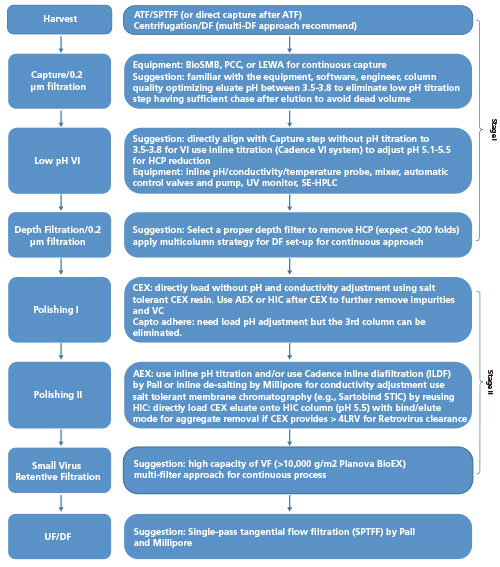
Antibody Purification Process Development and Manufacturing

Modeling Framework for Vaccine Production - BioProcess InternationalBioProcess International

Interpreting & working with protein chromatography chromatograms - w/practical look on an AKTA
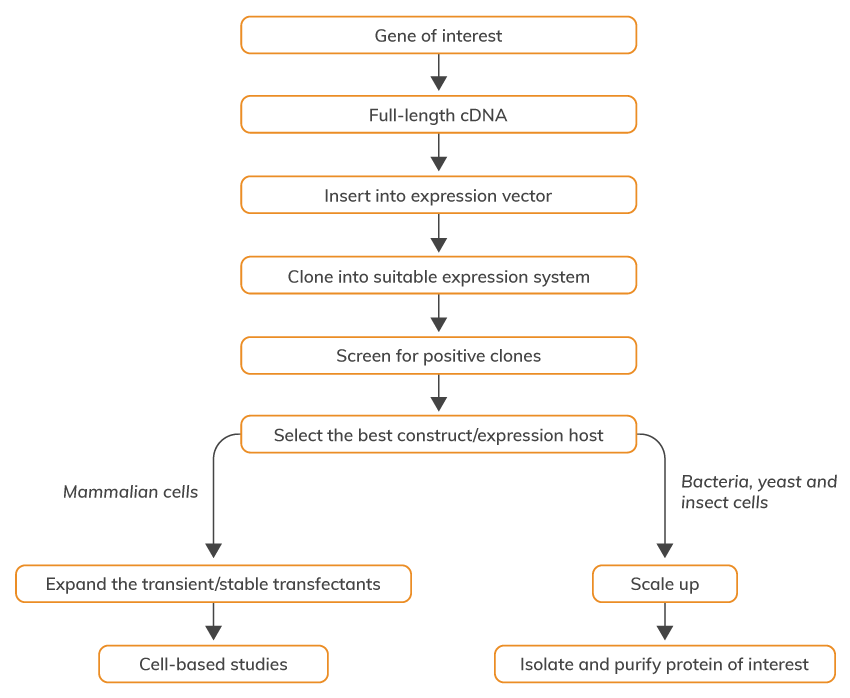
Learn about Recombinant Protein Production

Parallelized Protein Purification: Opportunities and Challenges in EarlyStage Biotherapeutics Research & Development
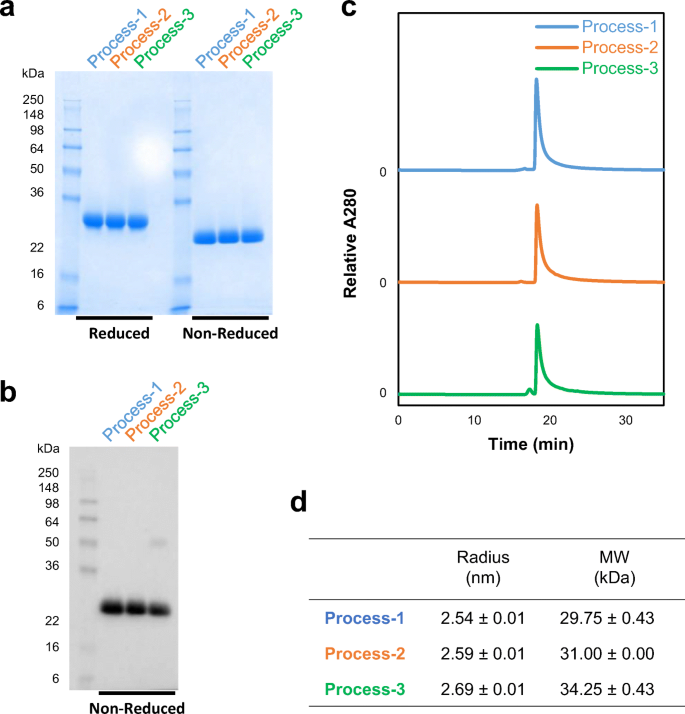
Process development and scale-up optimization of the SARS-CoV-2 receptor binding domain–based vaccine candidate, RBD219-N1C1

Development of a scalable and robust AEX method for enriched rAAV preparations in genome-containing VCs of serotypes 5, 6, 8, and 9: Molecular Therapy Methods & Clinical Development

Recent development and application of membrane chromatography

Development of a generic reversed-phase liquid chromatography method for protein quantification using analytical quality-by-design principles - ScienceDirect

Top-Down Proteomics of Large Proteins up to 223 kDa Enabled by Serial Size Exclusion Chromatography Strategy

Protein Chromatography: Process Development and Scale-Up 2, Carta, Giorgio, Jungbauer, Alois

MMMM, PDF, Chromatography




