Description and Appraisal of Outcome Measures - Clinical Review Report: Eculizumab (Soliris) - NCBI Bookshelf
Price: $ 222.99
4.6(693)
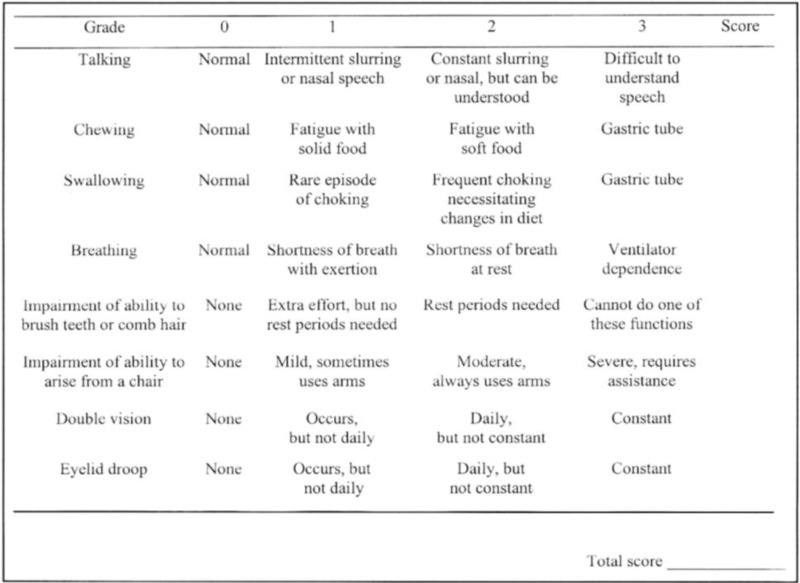
To describe the following outcome measures and review their measurement properties (validity, reliability, responsiveness to change, and MID): Myasthenia Gravis Activities of Daily Living scale (MG-ADL)Quantitative Myasthenia Gravis (QMG) scoreMyasthenia Gravis Composite (MGC) scoreMyasthenia Gravis Quality of Life 15-item scale (MG-QoL15)Myasthenia Gravis Foundation of America Post-Intervention Status (MGFA-PIS)Quality of Life in Neurological Disorders Fatigue Scale (Neuro-QoL Fatigue)European Quality of Life (EuroQol) Health 5-Dimensions (EQ-5D) questionnaire.

Full article: Quantifying the economic effects of ravulizumab

Clinical Evidence - Clinical Review Report: Eculizumab (Soliris
Clinical Evidence - Clinical Review Report: Eculizumab (Soliris
Hematopoietic stem cell transplantation-associated thrombotic

Costs and Utilization of New-to-Market Neurologic Medications

Clinical Evidence - Clinical Review Report: Eculizumab (Soliris

Clinically important change for the FACIT-Fatigue scale in
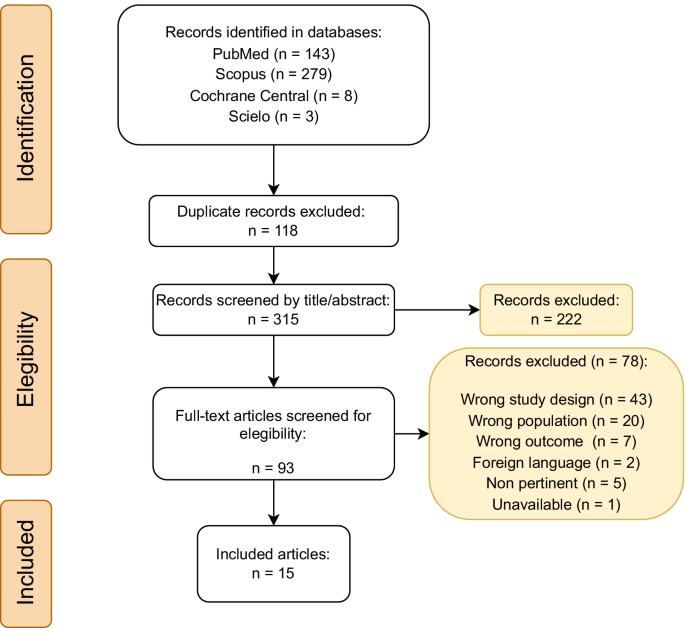
The treatment of atypical hemolytic uremic syndrome with
Clinical Evidence - Clinical Review Report: Eculizumab (Soliris

Description and Appraisal of Outcome Measures - Clinical Review

Eculizumab monotherapy for NMOSD: Data from PREVENT and its open

Brain Sciences, Free Full-Text
Ravulizumab: A Review in Atypical Haemolytic Uraemic Syndrome
Economic Review - Pharmacoeconomic Report: Eculizumab (Soliris




