
FDA approves first-of-its-kind “living drug” for childhood
Price: $ 65.00
4.5(701)
The Food and Drug Administration on Wednesday approved a groundbreaking cancer treatment for childhood leukemia that uses patients' own genetically altered immune cells to fight the disease. It is the first gene therapy to be allowed in the United States - a "historic" approval, the agency said.

Fam-trastuzumab deruxtecan-nxki active in early HER2-low breast cancer

FDA approves Dupixent® (dupilumab) as first biologic medicine for children aged 6 to 11 years with moderate-to-severe atopic dermatitis
/cloudfront-us-east-2.images.arcpublishing.com/reuters/G3FFOTIHGJJBBMLHXOA3PEU6VU.jpg)
U.S. FDA grants full approval to Pfizer COVID vaccine for ages 12-15

FDA approves semaglutide for obesity in adolescents

USFDA approves first Covid-19 treatment for young children

FDA approves 1st drug to delay onset of Type 1 diabetes - ABC News

Federal Register :: Authorizations of Emergency Use of Certain Drug and Biological Products During the COVID-19 Pandemic; Availability
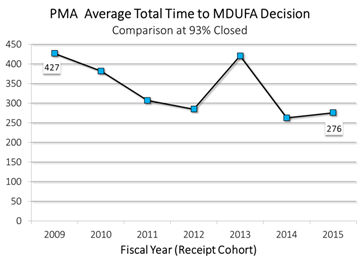
Prescription Drug User Fee Act Reauthorization (PDUFA VI), Medical Device User Fee Act Reauthorization (MDUFA IV), Generic Drug User Fee Act Reauthorization (GDUFA II), and Biosimilar User Fee Act Reauthorization (BsUFA II)
Coronavirus: FDA approves first COVID-19 treatment for children younger than 12 – KIRO 7 News Seattle




