Pharmaceutics, Free Full-Text
Price: $ 42.99
4.8(532)
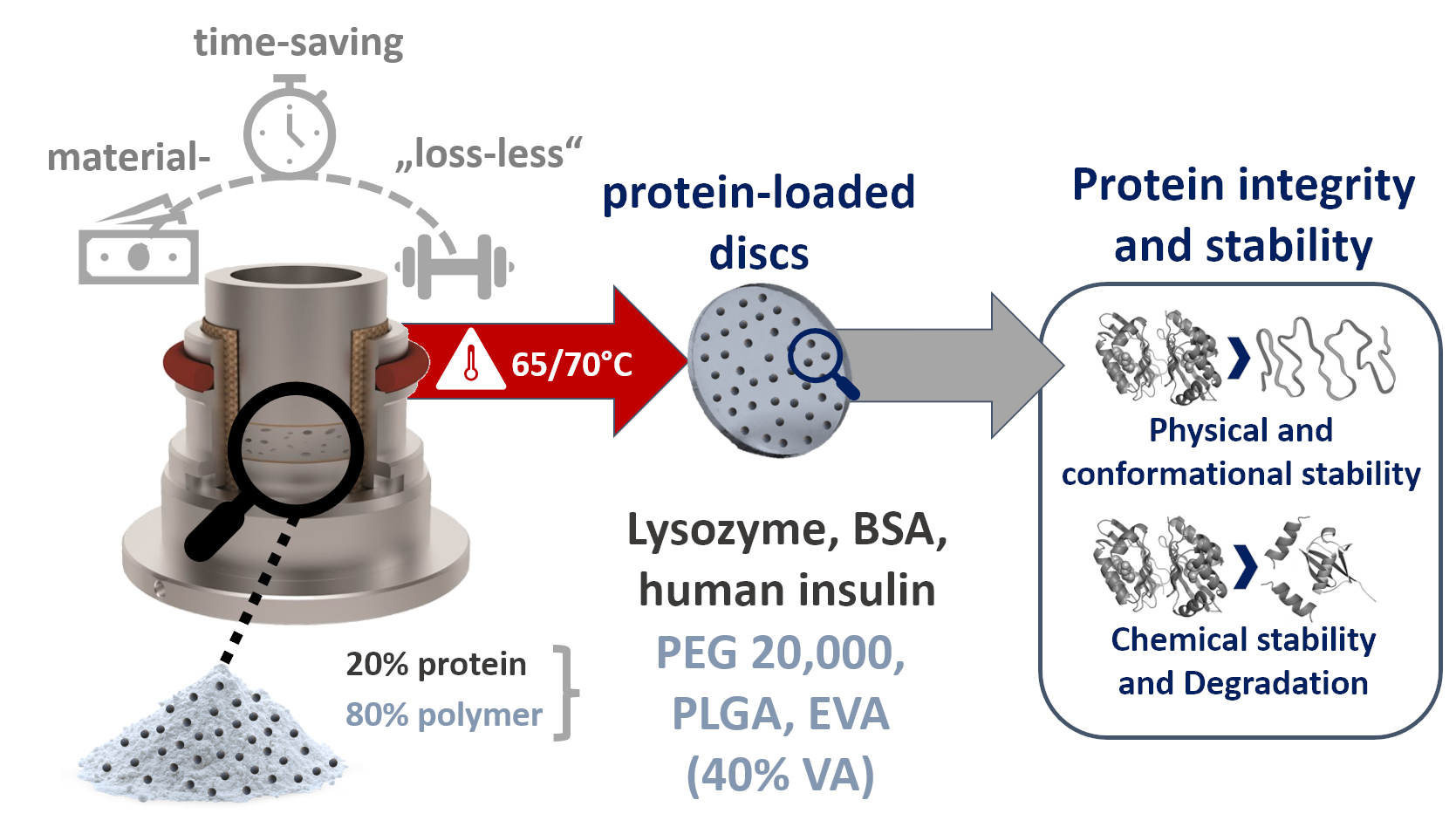
Hot-melt extrusion (HME) is used for the production of solid protein formulations mainly for two reasons: increased protein stability in solid state and/or long-term release systems (e.g., protein-loaded implants). However, HME requires considerable amounts of material even at small-scale (>2 g batch size). In this study, we introduced vacuum compression molding (VCM) as a predictive screening tool of protein stability for potential HME processing. The focus was to identify appropriate polymeric matrices prior to extrusion and evaluation of protein stability after thermal stress using only a few milligrams of protein. The protein stability of lysozyme, BSA, and human insulin embedded in PEG 20,000, PLGA, or EVA by VCM was investigated by DSC, FT-IR, and SEC. The results from the protein-loaded discs provided important insights into the solid-state stabilizing mechanisms of protein candidates. We demonstrated the successful application of VCM for a set of proteins and polymers, showing, in particular, a high potential for EVA as a polymeric matrix for solid-state stabilization of proteins and the production of extended-release dosage forms. Stable protein-polymer mixtures with sufficient protein stability after VCM could be then introduced to a combination of thermal and shear stress by HME and further investigated with regard to their process-related protein stability.

Free Pharmaceutical Account Manager Resume - Download in Word
Cpc Keygen Get File - Colaboratory
Design and fashion enthusiasmPharmaceutics, Free Full-Text, chanel
Pharmaceutical Services Flyer - PSDPixel

Pharmaceutics An Open Access Journal from MDPI
High reprint orders in medical journals and pharmaceutical
Pharmaceutics, Free Full-Text, thomas mitchell overton execution

RSC Pharmaceutics journal

MXenes and MXene-based materials for removal of pharmaceutical
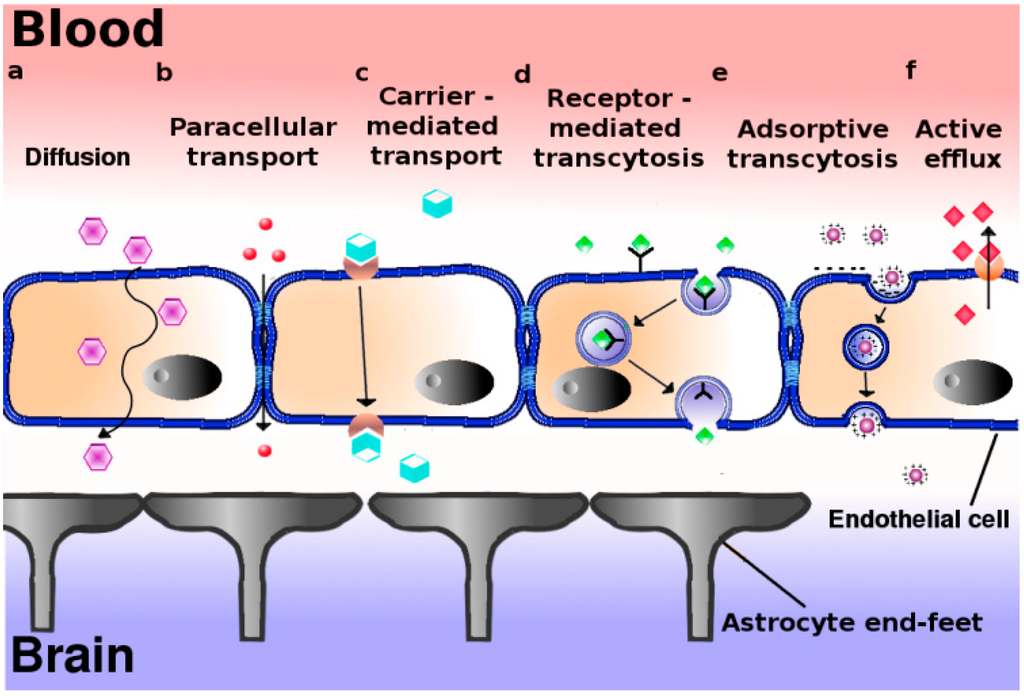
Pharmaceutics, Free Full-Text
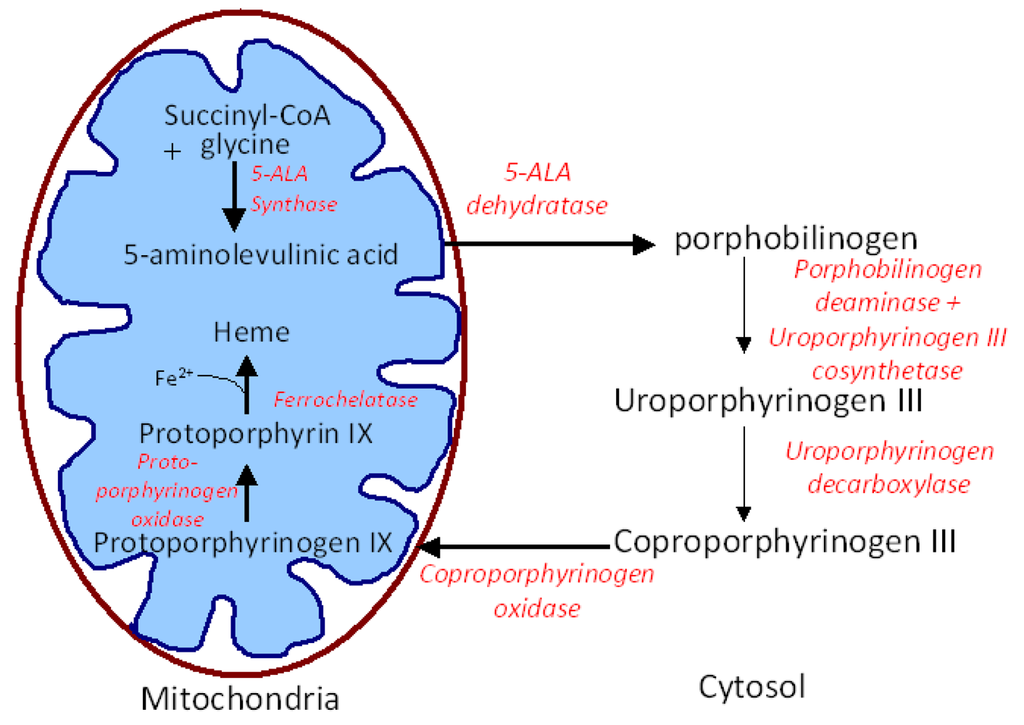
Pharmaceutics September 2011 - Browse Articles




