
Ice - Structure, Formation, Properties
Price: $ 7.50
4.8(326)
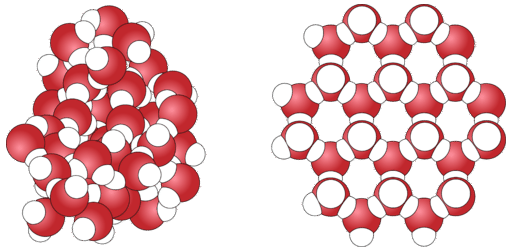
Ice - Structure, Formation, Properties: At standard atmospheric pressure and at temperatures near 0 °C, the ice crystal commonly takes the form of sheets or planes of oxygen atoms joined in a series of open hexagonal rings. The axis parallel to the hexagonal rings is termed the c-axis and coincides with the optical axis of the crystal structure. When viewed perpendicular to the c-axis, the planes appear slightly dimpled. The planes are stacked in a laminar structure that occasionally deforms by gliding, like a deck of cards. When this gliding deformation occurs, the bonds between the layers break, and the hydrogen atoms involved in those.
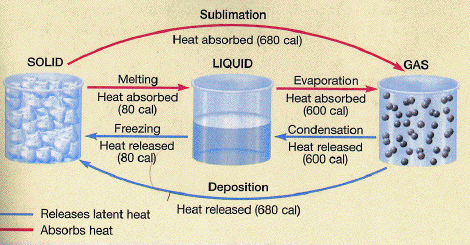
Ice, solid substance produced by the freezing of water vapour or liquid water. At temperatures below 0 °C (32 °F), water vapour develops into frost at ground level and snowflakes (each of which consists of a single ice crystal) in clouds. Below the same temperature, liquid water forms a solid, as
The Earth -- Introduction

The Crystalline Structure of Ice, Journal of Glaciology
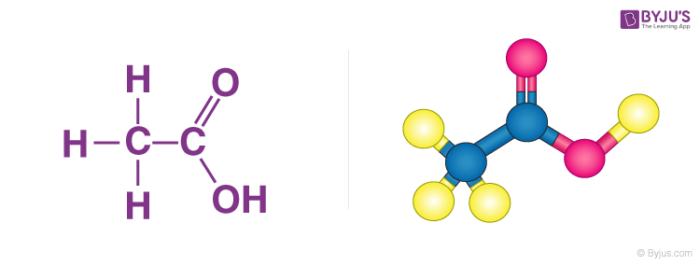
Acetic Acid (CH3COOH)- Structure, Properties, Preparation, Physical, Chemical properties, Uses and FAQs of Acetic acid

Ice - Wikipedia

Antarctic Landfast Sea Ice: A Review of Its Physics, Biogeochemistry and Ecology - Fraser - 2023 - Reviews of Geophysics - Wiley Online Library

Effect of Heat Treatment on Structure and Properties of Martensite Stainless Steel for Ice-Class Ships Obtained by Direct Laser Deposition Method
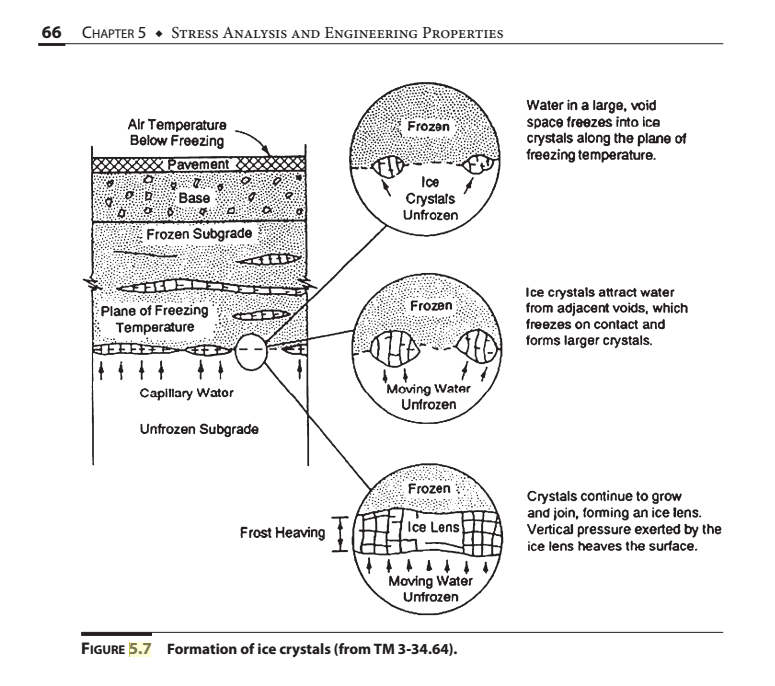
Solved 256 CHAPTER 11 • SHALLOW FOUNDATIONS AND PAVEMENTS
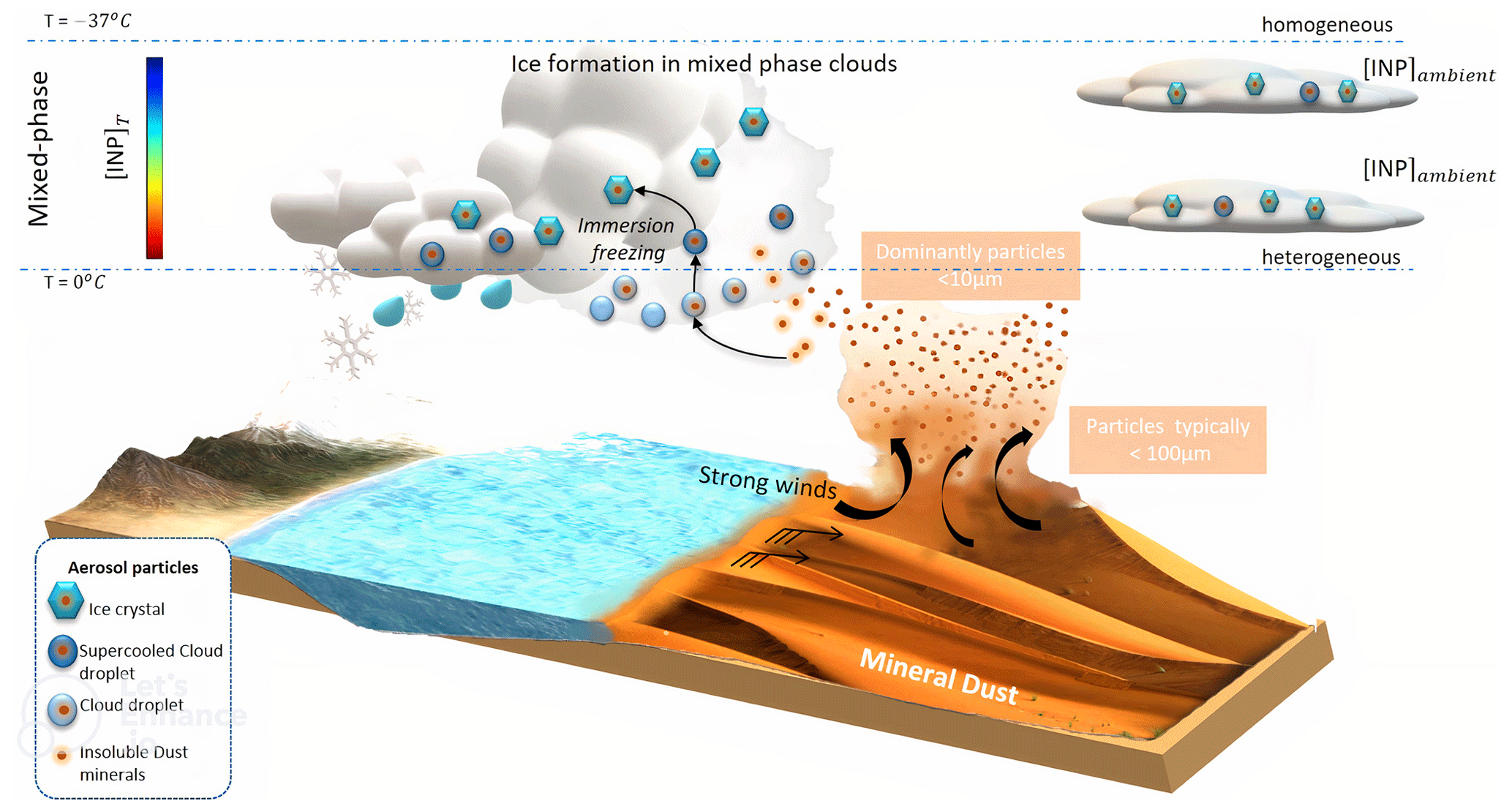
ACP - Role of K-feldspar and quartz in global ice nucleation by mineral dust in mixed-phase clouds
Lesson 10: Properties of Water

Describing the Properties of Ionic Solids, Chemistry
15.2: Structure of Ice - Chemistry LibreTexts




