
HSA Draft Guidance on Clinical Evaluation: Definitions
Price: $ 48.50
4.5(159)
The document describes in detail the aspects to be taken into account when preparing a site master file.

Background characteristics of the interviewed HSAs currently working in

Pharmaceuticals, Free Full-Text

HSA Guidance on Change Notification: Overview
HSA Draft Guidance on Clinical Evaluation: Definitions

MicroRNA hsa-miR-320a-3p and Its Targeted mRNA FKBP5 Were Differentially Expressed in Patients with HIV/TB Co-Infection

FDA Guidance on Reprocessing Medical Devices: Validation of Cleaning
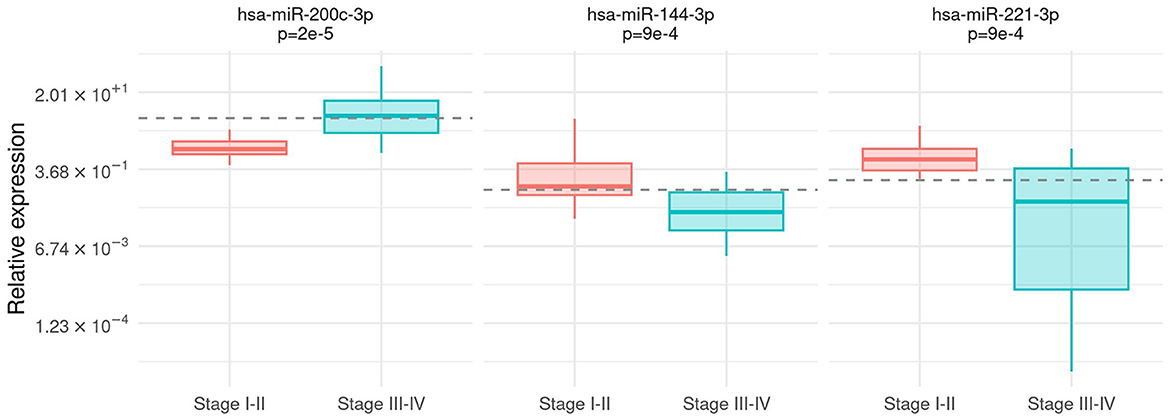
Frontiers A group of three miRNAs can act as candidate circulating biomarkers in liquid biopsies from melanoma patients

The Singapore Guidance on Software Medical Devices

HSA Consultation on Risk Classification of Standalone Medical Mobile Applications (SaMD)

Your Clinical Decision Support Software: Is It a Medical Device?
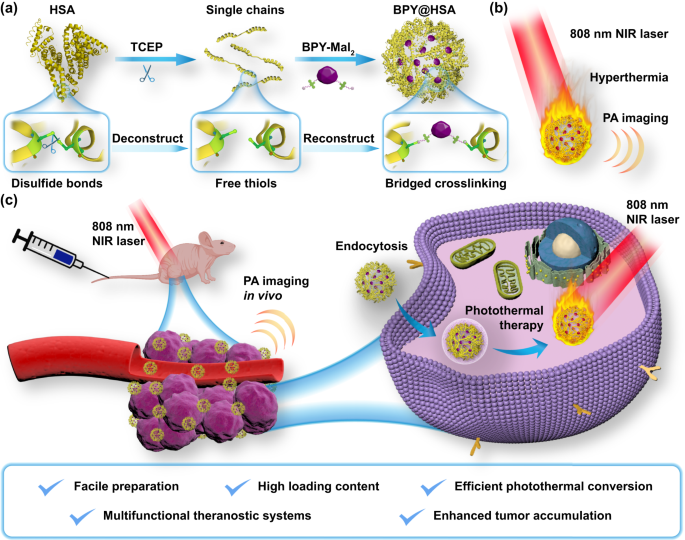
NIR-dye bridged human serum albumin reassemblies for effective photothermal therapy of tumor
Overexpression of hsa_circ_0002874 promotes resistance of non-small cell lung cancer to paclitaxel by modulating miR-1273f/MDM2/p53 pathway

HSA Guidance on Clinical Evaluation: Overview
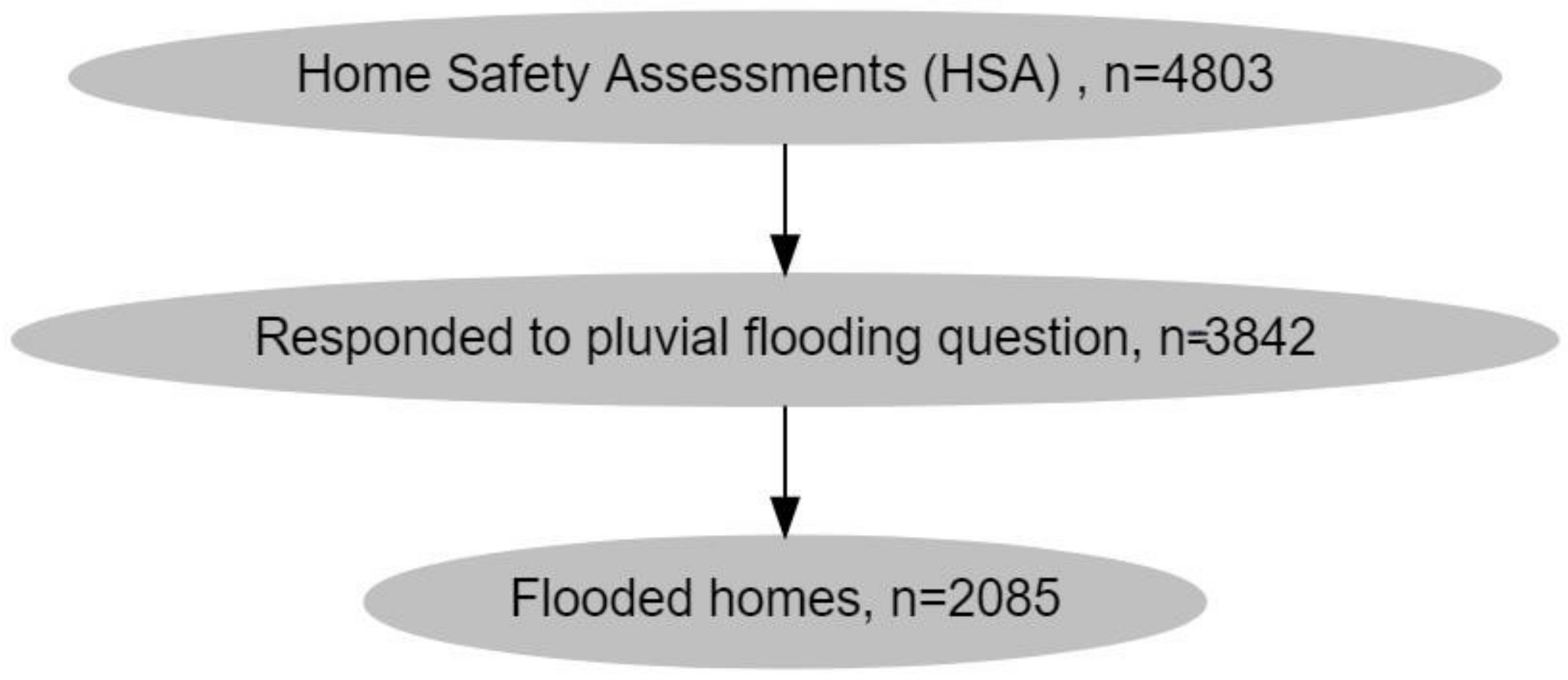
IJERPH, Free Full-Text




