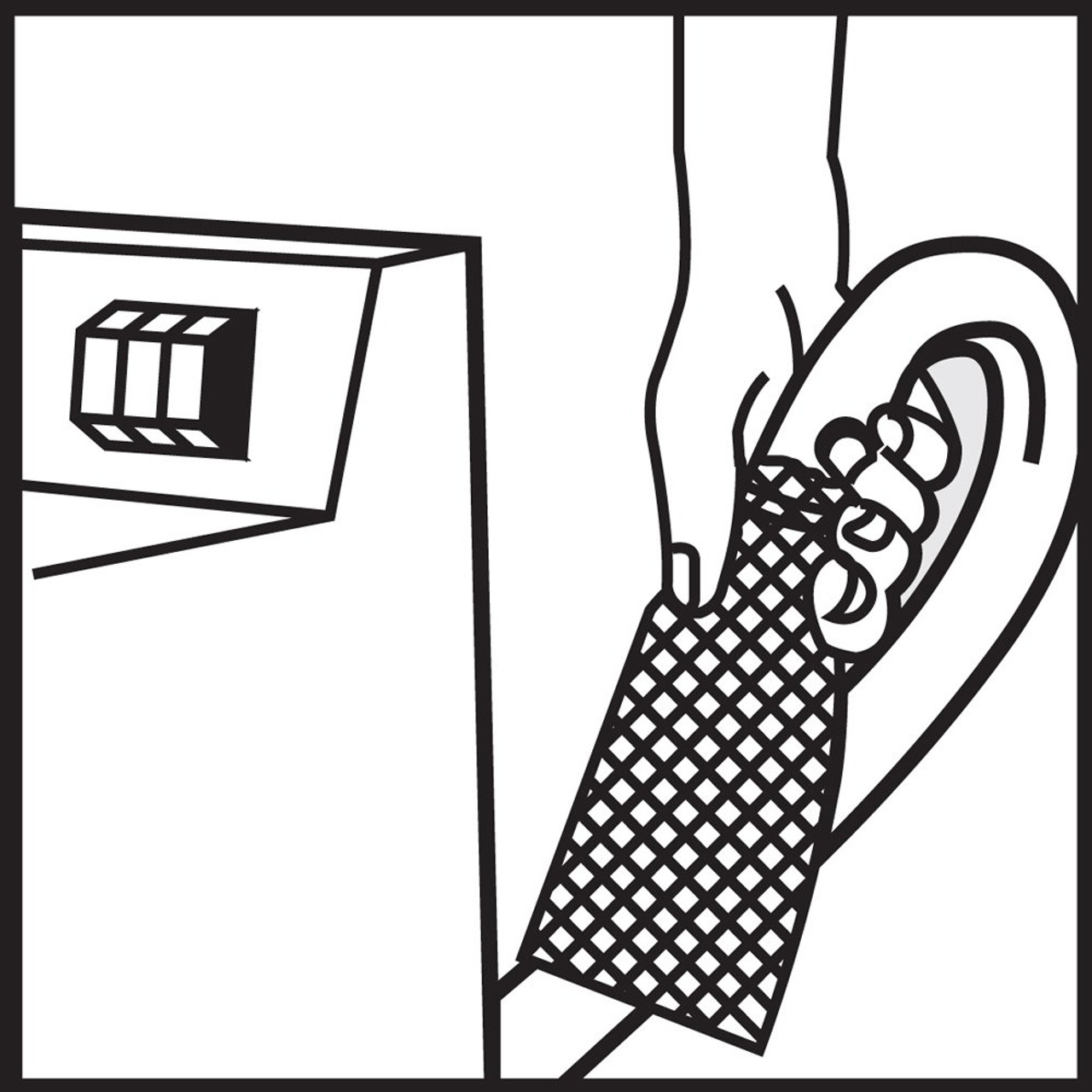
FDA gives nod to first fully-removable percutaneous peripheral
Price: $ 87.00
4.8(467)
SPR Therapeutics has developed a peripheral nerve stimulation system that is placed percutaneously through the skin instead of being implanted and which can be completely removed from the body after therapy period.

Nevro Announces FDA Approval of its 10 kHz High Frequency Spinal Cord Stimulation Therapy for Treatment of Chronic Pain Associated with Painful Diabetic Neuropathy (PDN)
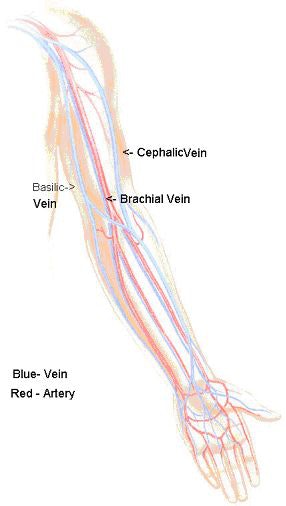
PICCing sides: Interventional radiologists weigh IV access lines

Nanomaterials, Free Full-Text

FDA approves Nevro's Senza system to treat chronic pain with diabetic neuropathy - MassDevice

A Surgeon's Guide for Various Lung Nodule Localization Techniques and the Newest Technologies - Katie N. Cornella, Danielle C. Repper, Brian A. Palafox, Mahmood K. Razavi, Christopher T. Loh, Kelly M. Markle

AHDB March/April 2015 Vol 8 Payor's Guide by Dalia Buffery - Issuu
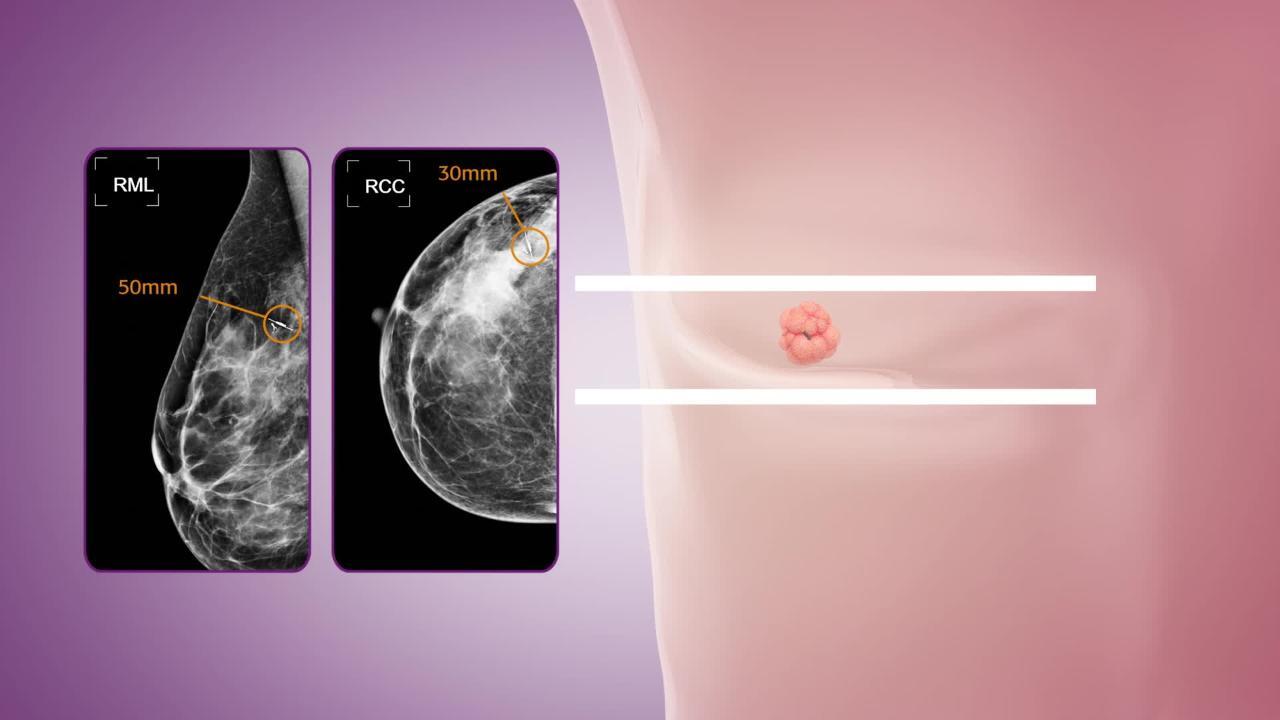
A Surgeon's Guide for Various Lung Nodule Localization Techniques and the Newest Technologies - Katie N. Cornella, Danielle C. Repper, Brian A. Palafox, Mahmood K. Razavi, Christopher T. Loh, Kelly M. Markle
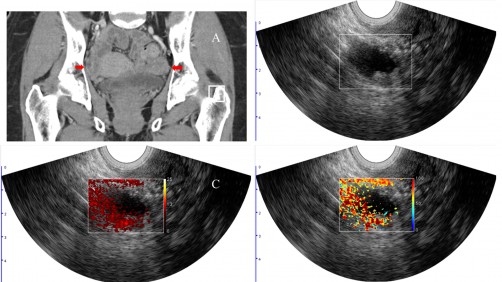
News - Interventional News

NEUSPERA MEDICAL® ANNOUNCES FDA CLEARANCE OF ITS SYSTEM FOR PERIPHERAL NERVE STIMULATION
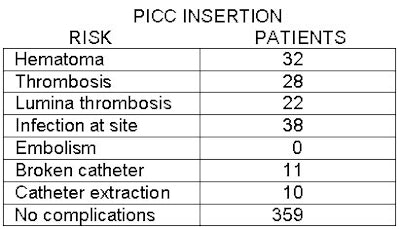
PICCing sides: Interventional radiologists weigh IV access lines

Advanced Healthcare Materials: Early View

Interventional News Issue 85—April 2022 Edition by BIBA Publishing - Issuu

NeuroNews issue 50 - US by BIBA Publishing - Issuu

Parkinson Disease: Translating Insights from Molecular Mechanisms to Neuroprotection

FDA gives nod to first fully-removable percutaneous peripheral nerve stimulation device - MedCity News