FDA Awards Fast Track Designation to Multiple Non-COVID-19
Price: $ 69.99
4.7(511)
While the world has largely been focused on the development of vaccines and therapeutics for COVID-19, the U.S. FDA has remained busy lining up potential approvals of medications for other diseases and illnesses.

Moderna's next act: more mRNA treatments after its COVID-19 vaccine

HRSA Says COVID Flexibility About Dispensing 340B Drugs in

Sellas presses ahead with fast track designation for lymphoma

FDA efficiency for approval process of COVID-19 therapeutics

Alliance Celebrates the First-Ever, FDA Traditional Approval of a

An overview of PRIME, Breakthrough Therapy & Fast Track procedures

2023 drug approvals: After a down year, FDA signs off on a bounty

The 'very, very bad look' of remdesivir, the first FDA-approved

FDA Grants Accelerated Approval to Elrexfio for Multiple Myeloma
Memo nabs fast track designation for BK polyomavirus drug
FDA Grants Breakthrough Device Designation to Anumana's ECG

Non-Small Cell Lung Cancer (NSCLC) Archives - DelveInsight
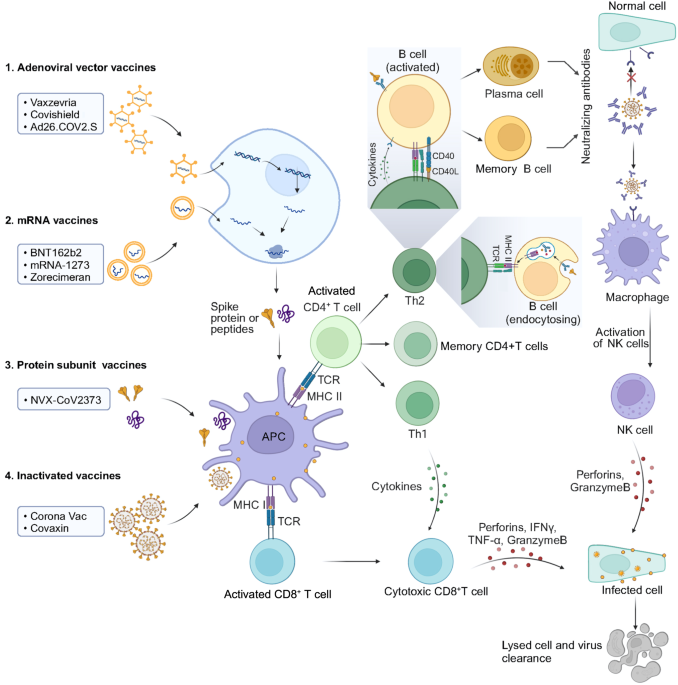
Strategies for the development and approval of COVID-19 vaccines

COVID-19: The inside story of the fastest vaccine ever developed




